סיכום קצר: Digital transformation in life sciences involves integrating AI, data analytics, telemedicine, and digital health technologies across drug development, clinical trials, manufacturing, and patient care. Only 20% of biopharma companies are digitally maturing, and the sector lags behind other industries despite AI initiatives. Success requires coordinated digital infrastructure, improved data quality, and strategic alignment with regulatory frameworks.
The life sciences industry stands at a crossroads. Digital technologies promise faster drug discovery, personalized medicine, and improved patient outcomes. But here’s the thing—most companies aren’t there yet.
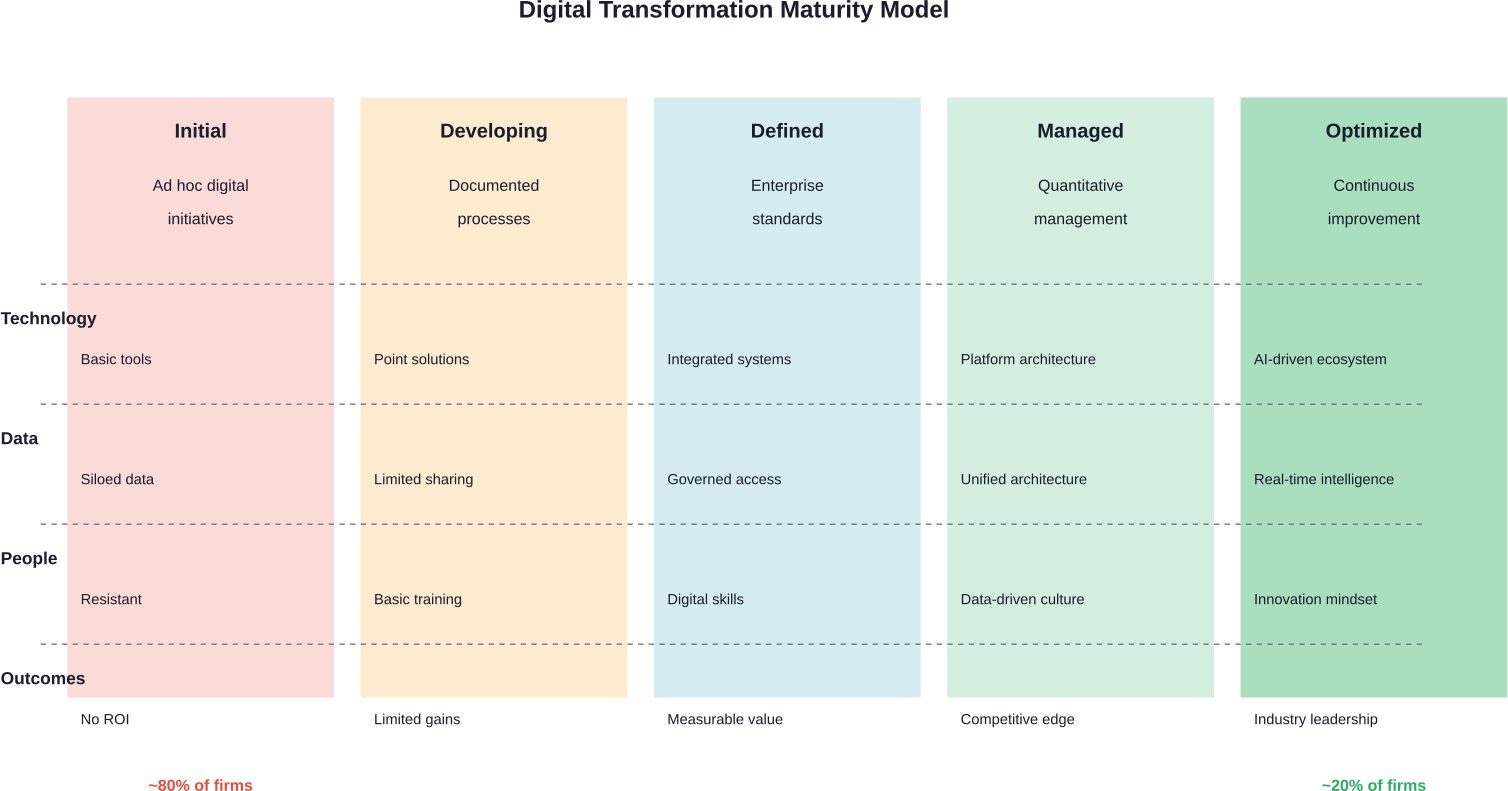
Only about 20 percent of biopharma companies have reached digital maturity. That’s a staggering gap considering the pace of innovation happening elsewhere. While AI can analyze thousands of molecular structures in hours and wearable devices continuously monitor patient health, many life sciences organizations still rely on paper-based processes and fragmented systems.
The transformation isn’t optional anymore. It’s a strategic imperative.
What Digital Transformation Means in Life Sciences
Digital transformation goes beyond installing new software. It’s about fundamentally changing how pharma and medtech companies operate, make decisions, and deliver value.
According to the FDA, Artificial Intelligence refers to machine-based systems that make predictions, recommendations, or decisions for real or virtual environments. These systems perceive environments, abstract perceptions into models through automated analysis, and use model inference to formulate options for action.
But transformation extends far beyond AI alone. It encompasses electronic medical records, telemedicine platforms, data-driven surveillance systems, and digital biomarkers that can detect disease earlier than traditional methods.
The WHO emphasizes that digital health applications remain largely untapped globally, with immense scope for solutions that can improve population health. Digital technologies are rapidly becoming integral to daily life, yet their application to health systems—particularly in low- and middle-income countries—faces significant coordination challenges.
From Doing Digital to Being Digital
Many life sciences companies are stuck in the “doing digital” phase. They launch pilot projects, adopt point solutions, and experiment with new technologies. That’s progress, but it’s not transformation.
Being digital means embedding technology into organizational DNA. Data flows seamlessly across departments. Decisions happen in real-time based on analytics. Patient insights shape R&D priorities from day one.
The shift requires cultural change, not just technical upgrades.

Key Technologies Driving Change
Several technologies are reshaping the life sciences landscape right now. Let’s break down the ones making the biggest impact.
בינה מלאכותית ולמידת מכונה
AI is accelerating drug discovery in ways that seemed impossible a decade ago. Research shows that 31% of life sciences companies report high or very high ROI from AI initiatives.
The global AI pharmaceutical market continues expanding rapidly. Machine learning algorithms can predict which molecular compounds might become effective drugs, analyze patient data to identify disease patterns, and optimize clinical trial designs.
But here’s where it gets tricky. Data quality matters enormously. Using datasets with an 80% accuracy rate may suffice for day-to-day business tasks, but it’s wholly insufficient for clinical applications. Building internal sensitivity to data quality becomes critical when lives depend on algorithmic decisions.
Digital Health Technologies and Wearables
Wearable technologies and smartphone applications now provide continuous health monitoring. A study of 3,246 people demonstrated that smartwatch-based alerting systems could detect pre-symptomatic COVID-19 signals up to three days before symptom onset in 78% of cases.
This changes everything about clinical research. Traditional site visits might capture 50 hours of participant data per month. Digital tools collecting data passively throughout the day can capture hundreds of hours of real-world evidence.
The WHO Global Strategy on Digital Health emphasizes that wearables facilitate early symptom detection and prompt intervention, making health systems more efficient and sustainable.
Real-World Evidence and Digital Biomarkers
Real-world evidence gathered from electronic health records, insurance claims, and patient registries is transforming regulatory science. As of April 2025, ClinicalTrials.gov lists 29% of registered studies with U.S. locations and 56% with international locations, reflecting the globalization of clinical research.
Digital biomarkers—objective, quantifiable physiological measures collected through digital devices—offer unprecedented insights into patient health between clinical visits. They’re making virtual and decentralized trials more feasible.
Advance Innovation in Life Sciences
Digital transformation in life sciences enables better research, improved healthcare services, and more efficient operations. Modern technology helps organizations manage data, accelerate innovation, and improve collaboration.
- Develop secure platforms for research and healthcare data
- Implement data analytics and AI solutions
- Build digital systems for clinical and operational workflows
רשימת מוצרים א' provides development expertise to support digital innovation in life sciences organizations.
Transformation Across the Value Chain
Digital transformation touches every part of life sciences operations. Here’s where the impact shows up most.
Research and Development
Drug discovery timelines are compressing. AI models screen millions of compounds virtually before any lab work begins. Machine learning predicts which candidates will succeed in trials with improving accuracy.
The FDA recognizes increased AI use throughout drug development and across therapeutic areas. Regulatory frameworks are evolving to accommodate these innovations while maintaining safety standards.
Digital collaboration platforms let global research teams work together seamlessly. Scientists share data, insights, and results in real-time rather than waiting for quarterly meetings or conference presentations.
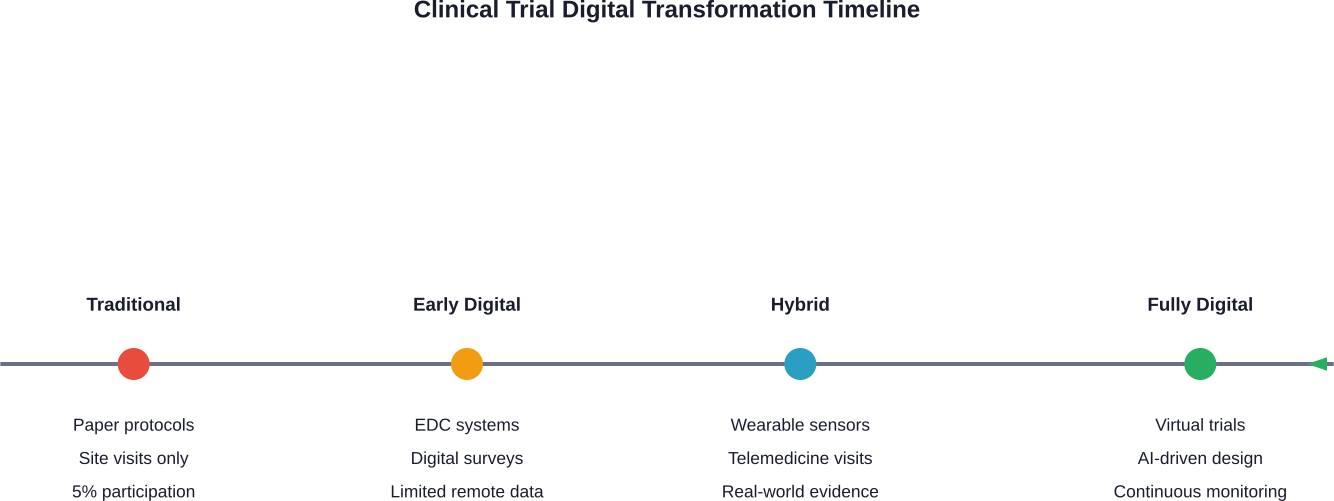
Clinical Trials Modernization
Only 5% of the U.S. population participates in clinical research. That’s a massive problem when developing treatments that work for diverse populations.
Digital tools are changing this equation. Virtual trials eliminate geographic barriers. Participants join from home using smartphones and wearable sensors. Digital surveys and remote monitoring make participation easier.
The result? Broader, more diverse participant pools. Faster enrollment. Better retention rates. More comprehensive data collection.
ייצור ושרשרת אספקה
Smart manufacturing uses IoT sensors, predictive maintenance, and real-time quality monitoring. Production becomes more efficient and compliant.
Supply chain visibility improves dramatically with digital tracking. Companies can monitor temperature-sensitive biologics throughout distribution, predict demand fluctuations, and respond to disruptions faster.
The pharmaceutical and medical device industries face different manufacturing challenges, but both benefit from digital process optimization and automated quality control systems.
Patient Engagement and Care Delivery
Telemedicine platforms connect patients with providers remotely. Mobile health apps help patients manage chronic conditions, track medications, and communicate symptoms.
Digital therapeutics—software-based interventions that treat medical conditions—are gaining regulatory approval. They’re not just health information apps; they’re prescribed treatments with clinical evidence behind them.
Patient portals give individuals access to their health records, test results, and treatment plans. This transparency improves engagement and outcomes.
התמודדות עם אתגרי היישום
Digital transformation sounds great in theory. Implementation is harder.
Data Integration and Quality
Life sciences companies often operate with siloed data systems. Research data lives separately from manufacturing data. Clinical trial results don’t connect easily with real-world evidence.
Creating unified data architectures requires significant investment and organizational change. Data governance policies need updating. Teams must agree on standards and definitions.
Data quality remains paramount. Clinical applications can’t tolerate the error rates acceptable elsewhere. Building systematic data quality checks becomes essential.
תאימות רגולטורית
Life sciences operates in heavily regulated environments. New technologies must comply with FDA requirements, EMA standards, and various national regulations.
Regulatory frameworks are evolving to address AI and digital health technologies, but gaps remain. Companies need clear guidance on validation requirements, data privacy protections, and approval pathways.
The WHO emphasizes that without strong national capacities to coordinate digital health efforts, transformation risks deepening inequalities rather than reducing them.
Skills and Organizational Culture
Digital transformation demands new skills. Data scientists, digital health specialists, and AI engineers become critical hires. Existing staff need training in digital tools and data-driven decision-making.
Cultural resistance poses real challenges. Clinicians accustomed to traditional methods may skeptically view digital interventions. Sales teams comfortable with in-person detailing must adapt to digital-first engagement models.
Change management becomes as important as technology selection.
| אזור האתגר | Common Obstacles | Strategic Solutions |
|---|---|---|
| אינטגרציית נתונים | Siloed systems, incompatible formats, legacy infrastructure | Unified data architecture, API-based integration, cloud migration |
| תאימות רגולטורית | Evolving standards, validation complexity, approval uncertainty | Early FDA engagement, robust documentation, quality-by-design |
| פער במיומנויות | Shortage of digital talent, insufficient training, resistance to change | Strategic hiring, continuous learning programs, cross-functional teams |
| ROI Measurement | Long timelines, difficult attribution, pilot-to-scale challenges | Clear KPIs, phased implementation, outcome-focused metrics |
Building a Successful Digital Strategy
What separates successful digital transformations from failed pilots? Strategy matters more than technology selection.
התחילו ביעדים ברורים
Don’t digitize for digitization’s sake. Define specific business outcomes. Faster drug development? Lower clinical trial costs? Better patient outcomes? Improved manufacturing efficiency?
Clear objectives guide technology choices and help measure success. They also build organizational buy-in by connecting digital initiatives to business priorities.
Take an Ecosystem Approach
Life sciences digital transformation can’t happen in isolation. Partnerships with technology vendors, academic institutions, and digital health startups accelerate progress.
Living Labs—collaborative environments where stakeholders co-create solutions in real-world settings—are gaining traction. These ecosystems bring together researchers, clinicians, patients, and technologists to drive innovation.
As noted in recent research, Living Labs facilitate digital health innovation through stakeholder collaboration and continuous iteration in actual healthcare environments.
Invest in Infrastructure
Digital transformation requires foundational infrastructure. Cloud computing platforms provide scalability. Data warehouses enable analytics. Interoperability standards allow systems to communicate.
The National Academy of Medicine emphasizes that the health sector continues lagging in developing robust digital health infrastructure, limiting potential gains in efficiency, access, and outcomes.
Infrastructure investment isn’t glamorous, but it’s essential. Without it, digital initiatives remain disconnected point solutions rather than integrated capabilities.
Prioritize Cybersecurity and Privacy
Healthcare data is incredibly sensitive. Breaches damage trust and trigger regulatory penalties.
Strong cybersecurity measures must be built into digital systems from the start, not added as afterthoughts. Encryption, access controls, audit trails, and incident response plans all become critical.
Privacy-preserving technologies like federated learning allow AI models to train on distributed datasets without centralizing sensitive information.
The Road Ahead
Digital transformation in life sciences isn’t a destination. It’s an ongoing journey as technologies evolve and new capabilities emerge.
Generative AI is already changing how scientists write protocols, analyze literature, and design molecules. Quantum computing promises breakthrough capabilities for molecular simulation. Edge computing will enable real-time analysis of wearable data without cloud transmission.
The companies that thrive will be those that build adaptable digital foundations rather than rigid systems. They’ll cultivate digital literacy across their organizations. They’ll partner strategically rather than trying to build everything in-house.
Most importantly, they’ll keep patients at the center. Technology serves no purpose if it doesn’t ultimately improve health outcomes and make care more accessible.
שאלות נפוצות
- What percentage of life sciences companies have achieved digital maturity?
Only about 20% of biopharma companies are considered digitally mature. The majority remain in earlier stages of transformation, still working on integrated systems and unified data architectures.
- What ROI can life sciences companies expect from AI initiatives?
According to industry research, 31% of life sciences companies report high or very high ROI from their AI initiatives. However, success depends heavily on data quality, clear objectives, and proper implementation.
- How are digital tools changing clinical trial participation?
Digital tools enable virtual and decentralized trials, eliminating geographic barriers. Traditional site visits might capture 50 hours of participant data monthly, while digital tools collecting data passively can capture hundreds of hours of real-world evidence.
- What are the biggest challenges to digital transformation in life sciences?
The main challenges include data integration across siloed systems, evolving regulatory requirements, skills gaps in digital talent, and organizational resistance to change. Data quality standards for clinical applications are particularly demanding.
- How is the FDA addressing AI in drug development?
The FDA recognizes the increased use of AI throughout drug development and across therapeutic areas. Regulatory frameworks are evolving to accommodate these innovations while maintaining safety standards, though guidance continues developing.
- What role do wearables play in digital health?
Wearables provide continuous health monitoring and enable early disease detection. Research showed that smartwatch-based systems could detect pre-symptomatic COVID-19 signals up to three days before symptom onset in 78% of cases. They facilitate real-world evidence collection and remote patient monitoring.
- Why is data quality so critical in life sciences digital transformation?
Clinical applications demand extremely high accuracy. Using datasets with an 80% accuracy rate may suffice for day-to-day business tasks, but it’s wholly insufficient for clinical applications. Poor data quality can lead to incorrect diagnoses, ineffective treatments, or regulatory failures.
התקדמות בתהליך הטרנספורמציה הדיגיטלית
The life sciences industry stands at a pivotal moment. Digital technologies offer unprecedented opportunities to accelerate discovery, improve patient outcomes, and deliver care more efficiently.
But capturing these opportunities requires more than technology purchases. It demands strategic vision, organizational commitment, and sustained investment in infrastructure, skills, and culture.
The 20% of companies that have reached digital maturity aren’t smarter or better funded. They’re more committed to comprehensive transformation rather than isolated pilots. They treat digital capabilities as core competencies, not IT projects.
For organizations beginning their transformation journey, the message is clear: Start with strategy, not technology. Define outcomes, not features. Build foundations, not point solutions. And always keep the end goal in sight—better health for the patients these innovations ultimately serve.
The digital future of life sciences is already here. The question isn’t whether to transform, but how quickly and effectively companies can adapt to remain competitive and relevant in an increasingly digital healthcare ecosystem.



