Quick Summary: Digital transformation in pharma leverages AI, cloud computing, IoT, and data analytics to accelerate drug discovery, optimize manufacturing, and personalize patient care. According to the Wyss Institute at Harvard University, AI-driven approaches have demonstrated potential to accelerate drug discovery, with examples like Insilico Medicine identifying a fibrosis treatment candidate in under 18 months. The sector is shifting from isolated pilot projects to enterprise-wide digital strategies that integrate operations, clinical trials, and supply chains.
The pharmaceutical industry faces pressure like never before. Development costs spiral upward, regulatory requirements intensify, and patient expectations shift toward personalized treatments.
Digital transformation isn’t just a buzzword anymore. It’s become the operating framework separating companies that lead from those struggling to keep pace.
But here’s the thing—only about 20 percent of biopharma companies are digitally maturing. The gap between early adopters and hesitant organizations widens every quarter.
The pandemic turbocharged digitalisation efforts. According to a GlobalData survey, nearly three-quarters of industry professionals agree that COVID-19 had the most significant impact on their digital initiatives, with 58% stating it accelerated transformation processes within their organizations.
What Digital Transformation Means for Pharma
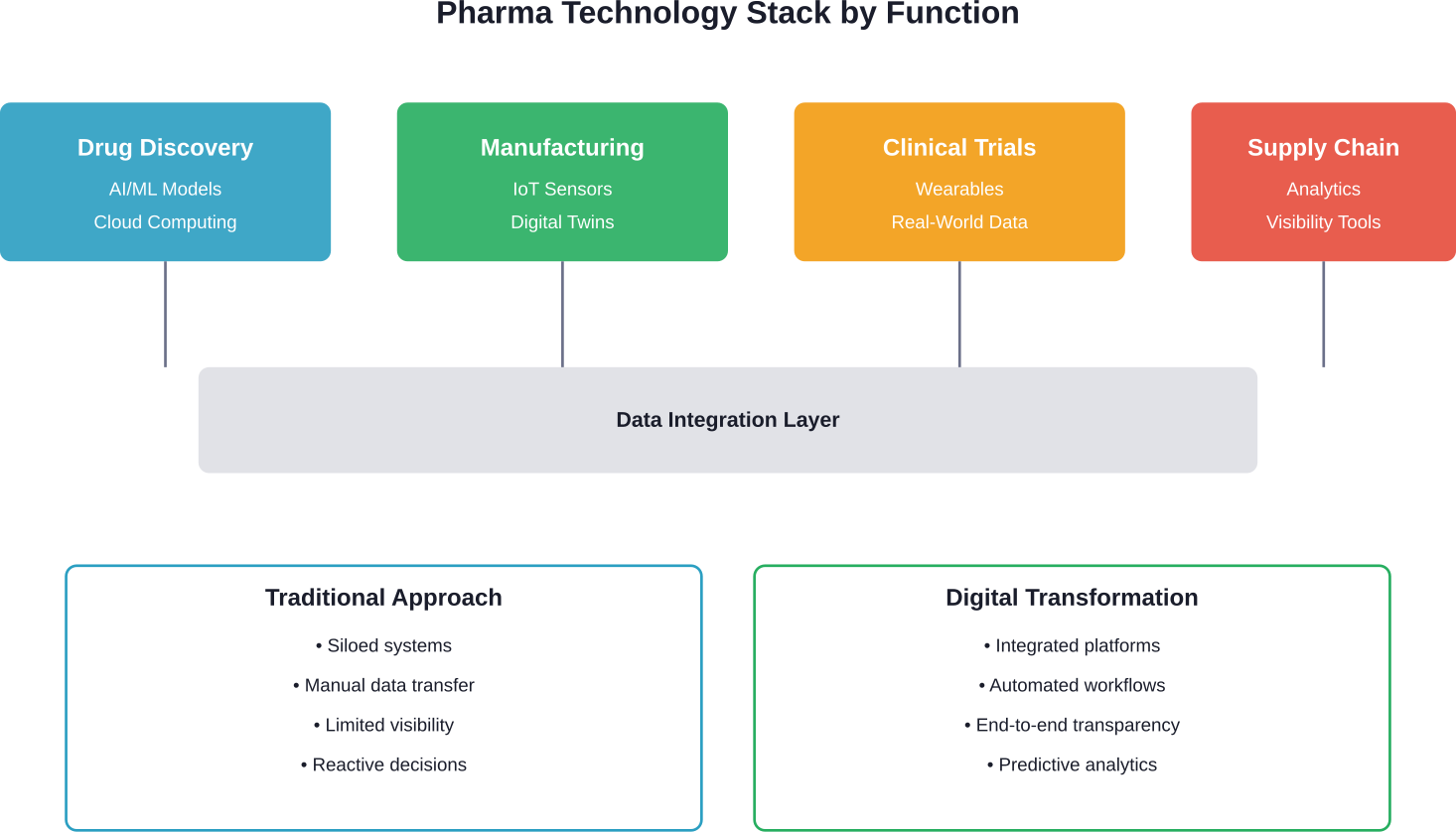
Digital transformation in the pharmaceutical industry goes beyond implementing new software. It’s a fundamental rewiring of how companies discover drugs, manufacture products, manage supply chains, and engage patients.
Traditional pharma operated in silos. R&D teams worked separately from manufacturing. Supply chain visibility extended only one tier deep. Clinical trial data sat disconnected from real-world evidence.
That model doesn’t cut it anymore.
Real digital transformation connects these pieces. Cloud platforms enable collaboration across continents. AI models screen millions of molecular combinations in hours rather than years. IoT sensors provide real-time visibility from raw materials to patient delivery.
According to the National Academy of Medicine’s recent paper on health digital architecture, the health sector continues to lag in developing robust digital infrastructure. This limits potential gains in efficiency, access, prevention, diagnosis, treatment, and discovery.
The pharmaceutical sector must address this gap to remain competitive.
Accelerate Pharma Innovation with Technology
Pharmaceutical companies rely on data platforms, research systems, and digital tools to manage complex operations and support innovation. Modern software solutions improve collaboration and data accessibility across teams.
- Develop secure platforms for research and data analysis
- Integrate data systems across departments
- Build scalable digital tools for operational workflows
A-listware supports pharma organizations with engineering teams and software development expertise for modern digital systems.
Core Technologies Reshaping the Pharma Value Chain
Several technologies drive meaningful change across pharmaceutical operations. Not every company needs every technology, but understanding the landscape helps prioritize investments.
Artificial Intelligence and Machine Learning
AI fundamentally changes drug discovery economics. According to the Wyss Institute at Harvard University, traditional drug discovery remains slow, expensive, and prone to high failure rates. Developing a new drug requires 13–15 years, with less than 10% of Phase I candidates receiving FDA approval, and the average R&D investment exceeds $2.5 billion when accounting for out-of-pocket expenses and abandoned trials.
In 2021, Insilico Medicine’s AI system identified a promising fibrosis treatment candidate in under 18 months—a timeline that typically spans years using conventional approaches. The AI model designed and validated a preclinical drug candidate in record time.
Beyond small-molecule development, in silico-based discovery extends to medicinal macromolecules. Researchers now design antimicrobial peptides, therapeutic proteins, and CRISPR-Cas9 systems using computational methods.
GlaxoSmithKline integrated AI across its laboratories, combining machine learning with automated robotics to screen compounds and predict biological activity. This isn’t a pilot project anymore—it’s core infrastructure.
Cloud Computing and Data Platforms
Cloud infrastructure solves the collaboration problem that plagued pharma for decades. Teams in Boston, Basel, and Bangalore can access the same datasets, run parallel experiments, and share findings in real time.
Cloud platforms also enable the elastic computing required for AI workloads. Training a drug discovery model might require massive computational resources for a week, then minimal resources afterward. Cloud economics make this feasible.
Data analytics platforms optimize R&D by connecting disparate information sources. Companies integrate clinical trial data, real-world evidence, genomic databases, and chemical libraries into unified analytics environments.
Internet of Things and Smart Manufacturing
IoT sensors transform pharmaceutical manufacturing from reactive to predictive. Temperature monitors, pressure gauges, and vibration sensors feed continuous streams of data into analytics platforms.
Digital twins—virtual replicas of physical production lines—let manufacturers test process changes without risking actual batches. If a temperature adjustment might improve yield, the digital twin simulates the outcome before implementation.
Smart manufacturing with IoT delivers measurable results. According to industry sources, companies deploying comprehensive digital manufacturing strategies report 1.75x higher operational equipment effectiveness compared to industry averages.
Real-World Data and Wearable Devices
Clinical trials historically depended on periodic clinic visits and patient-reported outcomes. Wearables and connected devices change this paradigm entirely.
Patients in trials now wear sensors that continuously monitor heart rate, activity levels, sleep patterns, and other biomarkers. This real-world data provides richer insights than traditional trial protocols.
Digital biomarkers enable personalized medicine at scale. Instead of treating every patient with the same protocol, physicians adjust treatments based on continuous feedback from wearables and connected devices.
Real-World Implementation Examples
Concrete examples show what’s actually working. Several pharmaceutical companies demonstrate measurable outcomes from digital investments.
Insilico Medicine represents the AI-driven discovery model. Their platform identified a fibrosis treatment candidate in under 18 months—a process that traditionally takes four to five years. The AI designed novel molecular structures, predicted their properties, and prioritized candidates for synthesis.
GlaxoSmithKline embedded AI throughout its research operations. The company doesn’t treat AI as a separate initiative but as integrated infrastructure. Automated labs screen compounds while machine learning models predict biological activity and potential side effects.
Smart manufacturing implementations deliver operational improvements. Companies deploying comprehensive IoT and digital twin strategies report operational equipment effectiveness 1.75 times higher than industry benchmarks.
When vulnerabilities appear in operational technology systems, integrated platforms automatically map them to affected equipment and production processes. The system prioritizes based on actual risk and schedules remediation during planned downtime rather than forcing emergency shutdowns.
The Pharma 4.0 Operating Model
ISPE’s Pharma 4.0 framework provides guidance for digital transformation efforts in pharmaceutical manufacturing and operations. The model identifies four aspects that require intentional management for successful transformation.
The framework moves beyond technology selection to address organizational change, data governance, workforce development, and continuous improvement processes.
Companies that treat digital transformation purely as technology implementation struggle. Those that address culture, skills, and operating models alongside technology see sustainable results.
| Technology | Primary Use Case | Typical Timeline |
|---|---|---|
| Cloud + AI | Vaccine and drug development | 12-18 months |
| Advanced Analytics | R&D optimization | 6-12 months |
| AI + Robotics | Molecule screening and autonomous labs | 18-24 months |
| Wearables + RWD | Personalized medicine and trials | 6-12 months |
| IoT + Digital Twins | Smart manufacturing | 12-18 months |
Challenges and Barriers
Digital transformation sounds compelling in presentations. Implementation reveals significant challenges.
Legacy systems create the first hurdle. Pharmaceutical companies operate manufacturing equipment, laboratory instruments, and enterprise software installed decades ago. These systems weren’t designed for integration.
Connecting legacy infrastructure to modern cloud platforms requires middleware, careful data mapping, and often custom integration work. It’s not impossible, but it takes time and specialized expertise.
Data governance presents another challenge. Pharmaceutical data must meet strict regulatory requirements. Companies can’t simply dump everything into a data lake and hope for compliance.
Robust data governance frameworks address data quality, lineage, access controls, and audit trails. The governance layer often requires as much attention as the technology layer.
Workforce skills represent a third barrier. Data scientists, machine learning engineers, and cloud architects don’t grow on trees. The competition for these professionals intensifies every year.
Many pharma companies address this through partnerships. Rather than building every capability in-house, they partner with technology providers, contract research organizations, and specialized consultants.

Strategic Priorities for 2026
Companies starting or accelerating digital transformation in 2026 should focus on specific priorities that deliver measurable value.
Start with problems, not technologies. The companies achieving the best results identify specific business challenges first, then select appropriate technologies. Starting with “we need AI” leads to solutions searching for problems.
Prioritize data infrastructure. Fancy algorithms don’t help if the underlying data remains fragmented, inconsistent, or inaccessible. Investing in data platforms, governance, and quality pays dividends across every subsequent initiative.
Build partnerships strategically. No company can develop every required capability in-house. Partnerships with technology providers, academic institutions, and specialized consultants accelerate progress.
Focus on workforce development. Technology alone doesn’t transform organizations—people do. Training programs, hiring strategies, and cultural initiatives determine whether new technologies get adopted or sit unused.
According to the National Academy of Medicine, developing robust digital health infrastructure requires coordinated investment across the health sector. Individual company efforts help, but industry-wide infrastructure development unlocks greater potential.
FAQ
- What does digital transformation mean in pharma?
Digital transformation in pharma refers to integrating technologies like AI, cloud computing, IoT, and data analytics across the entire value chain—from drug discovery through manufacturing to patient delivery. It’s not just implementing new software but fundamentally changing how companies operate, make decisions, and create value.
- Which technologies drive pharma digital transformation?
Core technologies include artificial intelligence for drug discovery, cloud platforms for collaboration and analytics, IoT sensors for smart manufacturing, wearable devices for real-world data collection, and digital twins for process optimization. The specific mix depends on company priorities and maturity level.
- How long does pharma digital transformation take?
Meaningful transformation typically spans three to five years. Pilot projects might show results in 6-12 months, but enterprise-wide integration, data governance, and cultural change require longer timelines. Only about 20 percent of biopharma companies have reached digital maturity, indicating most organizations remain in early stages.
- What ROI can pharma companies expect from digital transformation?
Companies implementing comprehensive digital manufacturing strategies report operational equipment effectiveness 1.75 times higher than industry averages. AI-driven drug discovery can compress development timelines from years to months. Specific ROI varies by initiative, but successful transformations typically show measurable improvements in speed, cost, and quality.
- What are the biggest challenges in pharma digital transformation?
Legacy system integration creates technical challenges. Data governance and regulatory compliance require careful framework development. Workforce skills gaps demand investment in training and strategic hiring. Cultural resistance to change affects adoption. Companies that address organizational and cultural aspects alongside technology achieve better outcomes.
- Do smaller pharma companies need digital transformation?
Yes. Digital technologies actually benefit smaller companies disproportionately by providing capabilities previously accessible only to large enterprises. Cloud platforms eliminate massive infrastructure investments. AI tools democratize advanced analytics. Strategic partnerships help smaller organizations access specialized expertise without building every capability in-house.
- How does Pharma 4.0 relate to digital transformation?
Pharma 4.0 is ISPE’s framework for guiding digital transformation in pharmaceutical manufacturing and operations. It identifies four key aspects that require intentional management: technology implementation, organizational change, data governance, and continuous improvement. The framework helps companies move beyond technology selection to address holistic transformation.
The Path Forward
Digital transformation for pharma isn’t optional anymore. The gap between digitally mature companies and laggards widens each quarter.
According to industry analysis, digital-native approaches are projected to capture significant market share by 2030, fundamentally transforming how drugs are developed. Companies that move now position themselves to lead with AI-driven discovery, real-time manufacturing optimization, and integrated clinical operations.
But remember—digital transformation succeeds when companies focus on business outcomes rather than technology for its own sake. Start with clear problems. Build solid data foundations. Invest in people alongside technology.
The pharmaceutical companies thriving five years from now won’t necessarily be those with the biggest technology budgets. They’ll be the organizations that integrated digital capabilities into their operating models most effectively.
The transformation journey continues. What matters is starting thoughtfully and progressing consistently toward measurable goals.



