Quick Summary: Digital transformation in cell and gene therapy leverages advanced manufacturing technologies, AI/ML platforms, digital twins, and integrated data systems to accelerate development timelines, improve product quality, enhance patient outcomes, and scale production from autologous to allogeneic therapies. According to FDA guidance, these innovations address critical manufacturing challenges while maintaining regulatory compliance and supply chain reliability.
Cell and gene therapies have shifted from experimental treatments to commercial realities. But scaling these personalized medicines presents unprecedented manufacturing, regulatory, and logistical challenges.
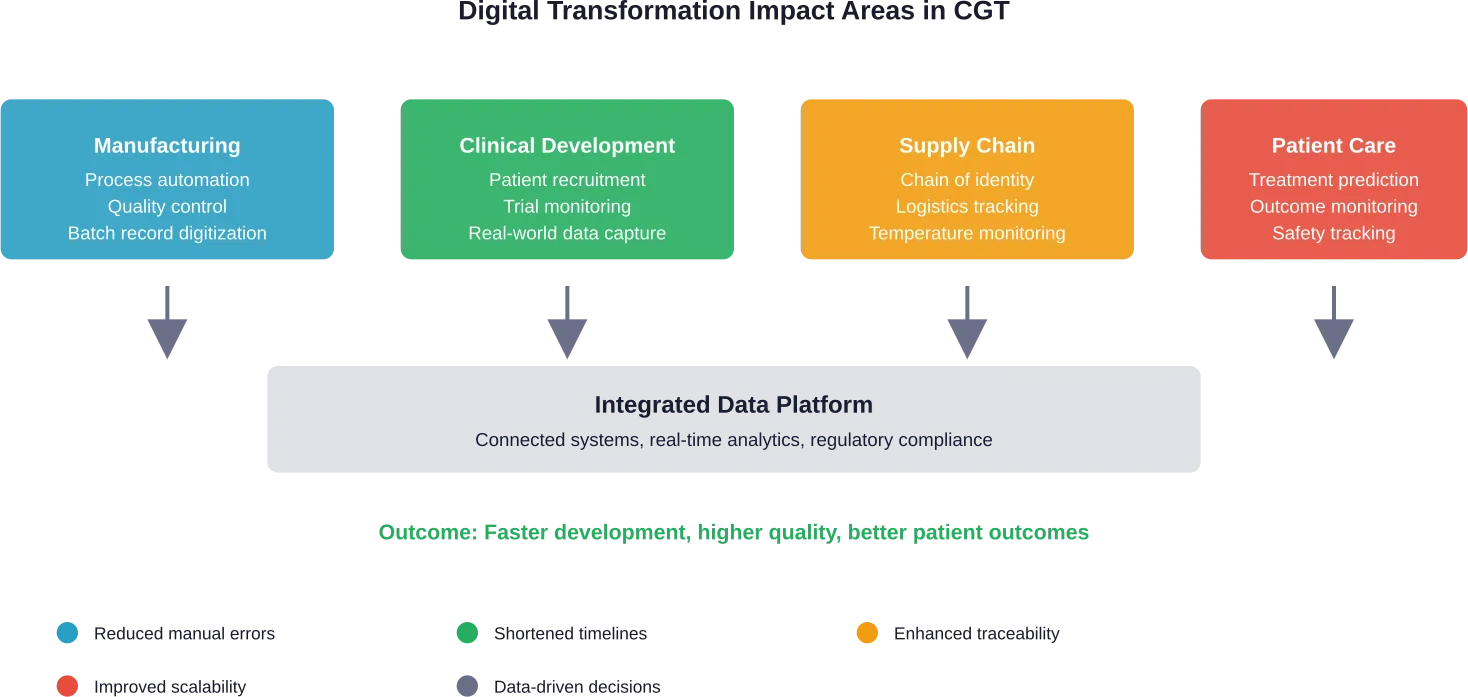
Digital transformation offers solutions. From AI-driven patient matching to digital twins predicting treatment response, technology is reshaping every stage of the CGT value chain.
Here’s how digital innovation is advancing cell and gene therapy development, manufacturing, and delivery—and what it means for patients and providers.
The CGT Market Landscape and Digital Imperatives
Global estimates project the cell and gene therapy market will reach $93.78 billion by 2030. This explosive growth creates urgent operational demands.
Traditional paper-based workflows can’t keep pace. Manual processes introduce errors, slow production cycles, and compromise data integrity—critical issues when manufacturing patient-specific therapies with tight timelines.
The FDA has recognized these challenges. Through its Advanced Manufacturing Technologies Designation Program, the agency encourages early adoption of technologies that improve manufacturing dependability and optimize development timelines for drug and biological products.
Digital transformation addresses core CGT challenges:
- Complex supply chain coordination across collection sites, manufacturing facilities, and treatment centers
- Maintaining chain of identity and chain of custody for autologous products
- Real-time quality monitoring and release testing
- Regulatory compliance documentation and audit trails
- Patient scheduling and vein-to-vein timeline management
The stakes are high. Treatment failure rates remain significant even with promising therapies—CD19-CAR therapy achieves remission rates of 70-90% in hematologic cancers, yet many patients eventually relapse due to antigen downregulation and tumor evasion mechanisms.
Advanced Manufacturing Technologies Reshaping Production
The FDA defines advanced manufacturing as “a collective term for new medical product manufacturing technologies that can improve drug quality, address shortages of medicines, and speed time-to-market.”
For CGT specifically, advanced manufacturing encompasses continuous processing, automation platforms, process analytical technology, and real-time quality monitoring systems.
Electronic Batch Records and Process Automation
Replacing paper-based batch records represents a foundational digital transformation step. Electronic batch records eliminate transcription errors, provide real-time process visibility, and create audit-ready documentation automatically.
Leading organizations report significant benefits:
- Reduced manufacturing cycle times through automated workflow transitions
- Improved data integrity with electronic signatures and timestamp verification
- Faster batch release through integrated quality review processes
- Enhanced collaboration between manufacturing sites and sponsors
But successful implementation requires more than software deployment. Organizations must redesign workflows, train personnel, and integrate systems across quality, manufacturing, and regulatory functions.
Chain of Identity and Chain of Custody Systems
Autologous therapies demand absolute certainty that the right product reaches the right patient. Digital chains of identity systems use biometric verification, RFID tracking, and barcode scanning to maintain product traceability from collection through infusion.
These systems integrate with scheduling platforms, logistics providers, and hospital information systems—creating end-to-end visibility while reducing manual verification steps that slow production timelines.
Process Analytical Technology and Real-Time Release
Traditional quality testing occurs at discrete batch endpoints. Process analytical technology enables continuous monitoring of critical quality attributes during production.
Real-time data collection supports faster decision-making and identifies deviations before they compromise product quality. Some advanced facilities are implementing real-time release testing—where continuous monitoring data replaces end-product testing, dramatically shortening release timelines.
Digital Twins and AI-Driven Treatment Optimization
Digital twin technology creates virtual replicas of biological systems—enabling simulation and prediction before physical intervention. For cell and gene therapy, digital twins operate at multiple scales.
Patient-Level Digital Twins
Research published in medical journals demonstrates digital twins can predict CAR-T cell therapy outcomes by integrating genomic data, imaging results, wearable sensor information, and clinical records into multiscale simulations.
These patient-specific models help clinicians:
- Predict treatment response based on individual tumor characteristics and immune profiles
- Optimize dosing strategies to balance efficacy against toxicity risks
- Identify patients most likely to benefit from specific therapy approaches
- Monitor post-treatment response and detect early relapse signals
Machine learning algorithms trained on historical patient data improve prediction accuracy over time. As datasets grow, digital twins become increasingly precise at forecasting individual treatment trajectories.
Manufacturing Process Digital Twins
Beyond patient care, digital twins model manufacturing processes. Virtual production simulations identify optimal process parameters, predict yield outcomes, and test process changes without consuming actual patient material.
This capability proves especially valuable during technology transfer—when moving processes between development and commercial manufacturing facilities or scaling from small to large production volumes.
Clinical Trial Innovation Through Digital Technologies
Digital technologies are transforming how clinical trials are designed, conducted, and monitored—particularly important given limited patient populations for many CGT indications.
Decentralized Trial Components
While CGT administration requires specialized facilities, digital tools enable remote patient monitoring, virtual consultations, and home-based sample collection where appropriate.
Wearable devices track safety parameters continuously rather than at discrete clinic visits. Patient-reported outcomes flow directly into trial databases through mobile applications. Telemedicine platforms enable frequent check-ins without travel burdens.
Real-World Data Integration
The American Society of Gene & Cell Therapy recently submitted comments supporting Health Level Seven Fast Healthcare Interoperability Resources (HL7 FHIR) standards for real-world data integration.
Real-world data captured from electronic health records, insurance claims, and patient registries supplements traditional clinical trial information—providing insights into long-term safety, effectiveness in broader populations, and comparative treatment outcomes.
The FDA is exploring frameworks to incorporate real-world evidence into regulatory decision-making, particularly for post-approval safety monitoring and label expansion considerations.
Adaptive Trial Designs
Digital data platforms enable adaptive trial designs that modify protocols based on accumulating evidence. For rare disease indications with limited patient populations, adaptive approaches maximize information gain while minimizing patient exposure to ineffective treatments.
Early-phase trials increasingly combine phase 1 and 2 objectives—focusing simultaneously on safety and preliminary efficacy. This streamlined approach is scientifically justified and ethically appropriate given urgent unmet needs.
Regulatory Considerations and Compliance Frameworks
Digital transformation must align with evolving regulatory expectations. The FDA has issued guidance on advanced manufacturing technologies, artificial intelligence in medical devices, and digital health technologies in clinical trials.
Advanced Manufacturing Technologies Designation
The FDA issued the final guidance for the Advanced Manufacturing Technologies Designation Program in December 2024. This framework allows manufacturers to request designation for specific technologies that show potential to improve product quality, reduce manufacturing risks, or address drug shortages.
Designated technologies receive enhanced FDA engagement—including meetings to discuss development plans, manufacturing assessments, and regulatory pathways. This proactive collaboration helps organizations implement novel technologies while maintaining regulatory compliance.
Data Integrity and System Validation
Digital systems must meet rigorous data integrity requirements. Electronic records need audit trails documenting all data creation, modification, and deletion. System access requires role-based controls and regular review.
Computer system validation demonstrates that digital platforms consistently perform as intended. Validation protocols document system requirements, testing procedures, and ongoing monitoring—creating evidence that systems maintain data integrity throughout their lifecycle.
AI and Machine Learning Oversight
As AI technologies become embedded in CGT development and manufacturing, regulatory frameworks are adapting. The FDA recognizes that AI/ML systems involve “complex and dynamic processes” requiring different oversight approaches than traditional software.
Key considerations include algorithm transparency, training data representativeness, performance monitoring in deployment, and change control procedures when models are updated based on new data.
Overcoming Implementation Barriers
Despite clear benefits, digital transformation faces obstacles. Understanding common barriers helps organizations develop mitigation strategies.
Cultural and Organizational Resistance
Shifting from paper-based workflows to digital systems requires cultural change. Personnel accustomed to traditional processes may resist new approaches—especially if training is inadequate or benefits aren’t clearly communicated.
Successful implementations prioritize change management:
- Involve end users in technology selection and workflow design
- Provide comprehensive training with hands-on practice opportunities
- Identify and empower digital champions within each functional area
- Celebrate early wins and share success stories broadly
- Address concerns transparently and adjust implementations based on feedback
System Integration Complexity
CGT organizations operate multiple specialized systems—manufacturing execution systems, laboratory information management systems, enterprise resource planning platforms, clinical trial management systems, and more.
Creating seamless data flow across these systems requires careful integration architecture. Application programming interfaces enable system-to-system communication, but integration projects demand significant technical resources and testing.
Many organizations adopt phased approaches—prioritizing highest-value integrations first rather than attempting comprehensive connection immediately.
Resource Constraints and ROI Uncertainty
Digital transformation requires upfront investment in software, infrastructure, consulting services, and personnel time. For smaller organizations or those developing therapies for ultra-rare diseases, resource constraints can delay or prevent adoption.
Building clear business cases helps secure funding. Quantifying expected benefits—reduced cycle times, lower error rates, improved yields, faster regulatory submissions—creates tangible ROI projections that justify investment.
Cloud-based software-as-a-service models reduce initial capital requirements compared to on-premise systems, making digital tools more accessible to organizations with limited budgets.
| Challenge Area | Common Barriers | Mitigation Strategies |
|---|---|---|
| Technology Selection | Overwhelming vendor landscape, unclear feature differentiation | Define requirements first, pilot before full commitment, seek peer references |
| Data Migration | Legacy data quality issues, format incompatibilities | Clean data proactively, establish migration validation protocols, accept staged approach |
| Regulatory Compliance | Uncertainty about validation requirements, audit readiness concerns | Engage quality/regulatory early, leverage vendor validation packages, document thoroughly |
| Staff Training | Time constraints, varying technical proficiency levels | Role-based training paths, super-user model, ongoing support resources |
| Vendor Management | Multiple vendor relationships, integration dependencies | Prioritize platforms with open APIs, establish clear governance, maintain vendor scorecards |
Global Accessibility and Health Equity Considerations
Digital transformation must consider global accessibility challenges. Cell and gene therapy translation to low- and middle-income countries faces significant barriers.
Between 1991 and 2008, only about 2% of the 274,000 global clinical trials were conducted in Africa despite the continent bearing substantial disease burden. Limited regulatory capacity, infrastructure gaps, and resource constraints impede access to advanced therapies.
Digital technologies offer partial solutions:
- Telemedicine platforms enable remote specialist consultations without expensive travel
- Cloud-based regulatory systems reduce local infrastructure requirements
- Shared manufacturing networks could serve multiple regions efficiently
- Digital training programs build local capability without requiring in-person expert travel
However, technology alone doesn’t address fundamental inequities in healthcare access, funding mechanisms, and industrial capacity. Digital transformation should complement—not substitute for—broader efforts to democratize advanced therapy access globally.
Future Directions and Emerging Technologies
Digital innovation in CGT continues accelerating. Several emerging technologies show particular promise.
Blockchain for Supply Chain Verification
Blockchain distributed ledger technology creates immutable records of product handling, storage conditions, and custody transfers. For autologous therapies requiring absolute chain of identity assurance, blockchain offers cryptographically verified traceability.
Early implementations demonstrate feasibility, though widespread adoption awaits standardization and integration with existing systems.
Advanced Analytics and Predictive Manufacturing
Machine learning models trained on historical manufacturing data can predict batch outcomes, identify process deviations before they impact quality, and recommend parameter adjustments to optimize yield.
As manufacturing datasets grow, predictive capabilities improve—potentially enabling lights-out manufacturing with minimal human intervention for routine production steps.
Synthetic Biology and Computational Design
Computational tools accelerate engineered cell therapy design. Rather than empirical trial-and-error, synthetic biology approaches use modeling to design genetic circuits, predict cell behavior, and optimize therapeutic constructs in silico before physical testing.
This computational design capability complements digital twins—together creating comprehensive virtual environments for therapy development and optimization.
Practical Implementation Roadmap
Organizations beginning digital transformation should follow structured approaches rather than ad-hoc technology adoption.
Step 1: Assess Current State
Document existing processes, systems, and data flows. Identify pain points, inefficiencies, and compliance risks. Evaluate digital maturity across manufacturing, quality, clinical, and commercial functions.
Step 2: Define Strategic Vision
Establish clear objectives aligned with business priorities. What specific outcomes does digital transformation need to achieve? Faster time-to-market? Improved product quality? Better patient outcomes? Lower production costs?
Prioritize use cases based on expected impact and implementation feasibility.
Step 3: Select Appropriate Technologies
Evaluate platforms against defined requirements. Consider integration capabilities, vendor stability, regulatory compliance features, scalability, and total cost of ownership.
Avoid technology-first approaches that select tools before understanding needs.
Step 4: Execute Focused Pilots
Test selected technologies in controlled scopes before enterprise-wide deployment. Pilots validate expected benefits, identify implementation challenges, and build organizational confidence.
Document lessons learned and refine approaches before scaling.
Step 5: Scale Successful Initiatives
Expand proven pilot projects across additional sites, products, or functions. Invest in training, change management, and ongoing support infrastructure.
Establish governance frameworks ensuring consistent deployment and preventing unauthorized system modifications.
Step 6: Optimize Continuously
Monitor performance metrics, gather user feedback, and refine processes iteratively. Digital transformation isn’t a one-time project—it’s an ongoing capability requiring continuous attention.
Stay current with emerging technologies and evolving regulatory expectations through industry associations, conferences, and peer networks.
| Implementation Phase | Typical Duration | Key Deliverables | Success Metrics |
|---|---|---|---|
| Assessment | 1-2 months | Current state documentation, gap analysis, maturity scoring | Comprehensive understanding of baseline capabilities |
| Strategy Development | 1-2 months | Vision statement, prioritized use cases, roadmap, business case | Executive alignment and funding approval |
| Technology Selection | 2-3 months | Requirements document, vendor evaluations, selection decision | Platform choices aligned with requirements |
| Pilot Execution | 3-6 months | Configured system, trained users, pilot results, lessons learned | Validated benefits, refined implementation approach |
| Scale Deployment | 6-18 months | Enterprise rollout, expanded training, integration completion | Adoption targets, performance improvements |
| Continuous Optimization | Ongoing | Performance dashboards, regular reviews, enhancement backlog | Sustained benefits, user satisfaction, evolving capability |
Build Reliable Digital Foundations for Cell and Gene Therapy
Cell and gene therapy work depends on accurate data, traceable processes, and systems that can handle both research and production without breaking continuity. A-listware supports organisations by examining how data, systems, and workflows are currently structured, then reorganising them to improve consistency and control.
This often includes strengthening how information is stored and accessed, reducing manual handoffs, and ensuring systems can support growth without creating gaps between stages of work. If your current setup makes it harder to maintain control or scale safely, contact A-listware to get a clear, practical view of how to move forward.
Measuring Digital Transformation Success
Quantifiable metrics demonstrate transformation value and guide ongoing improvement.
Manufacturing metrics include cycle time reduction, batch success rates, deviation frequency, and time-to-release. Leading organizations report 20-40% improvements in these areas following digitization.
Quality metrics track error rates, investigation timelines, audit findings, and inspection outcomes. Electronic systems typically reduce documentation errors by 60-80% compared to paper processes.
Clinical metrics measure patient recruitment rates, protocol deviation frequency, data quality scores, and monitoring efficiency. Digital tools can cut recruitment timelines by 30% or more.
Financial metrics include cost per batch, inventory carrying costs, labor productivity, and return on digital investment. Comprehensive transformations typically achieve positive ROI within 18-36 months.
Frequently Asked Questions
- What are the biggest challenges in digitally transforming cell and gene therapy manufacturing?
The primary challenges include system integration complexity across multiple specialized platforms, cultural resistance from personnel accustomed to paper-based workflows, resource constraints limiting upfront investment, regulatory compliance uncertainty around novel technologies, and data migration from legacy systems. Organizations overcome these barriers through phased implementations, strong change management, clear ROI demonstration, and early regulatory engagement.
- How do digital twins improve CAR-T cell therapy outcomes?
Digital twins create patient-specific virtual models by integrating genomic data, tumor characteristics, immune profiles, imaging results, and clinical history. These multiscale simulations predict individual treatment response, optimize dosing strategies, identify patients most likely to benefit, and enable early detection of relapse signals. Machine learning algorithms improve prediction accuracy as datasets expand, making digital twins increasingly valuable for personalizing therapy approaches.
- What regulatory guidance exists for advanced manufacturing technologies in CGT?
The FDA finalized guidance in December 2024 establishing the Advanced Manufacturing Technologies Designation Program. This framework allows manufacturers to request designation for technologies improving quality, reducing risks, or addressing shortages. Designated technologies receive enhanced FDA engagement including development meetings and regulatory pathway discussions. Additional guidance addresses AI/ML in medical devices, digital health technologies in trials, and data integrity requirements for electronic systems.
- How long does digital transformation typically take for cell and gene therapy organizations?
Timelines vary based on scope and organizational readiness. Initial assessment and strategy development require 2-4 months. Technology selection adds 2-3 months. Pilot projects run 3-6 months. Enterprise-scale deployment spans 6-18 months for comprehensive transformation. However, organizations should view this as continuous evolution rather than finite projects—ongoing optimization continues indefinitely as technologies and needs evolve.
- Can smaller biotech companies afford digital transformation initiatives?
Yes, though approaches differ from large pharmaceutical companies. Cloud-based software-as-a-service platforms reduce capital requirements compared to on-premise systems. Focused implementations targeting highest-value use cases deliver benefits without comprehensive enterprise deployments. Contract development and manufacturing organizations increasingly offer digital services, allowing small companies to access advanced capabilities without building internal infrastructure. Phased approaches spread costs over time while delivering incremental value.
- How does digitization improve supply chain management for autologous therapies?
Digital supply chain platforms integrate collection site scheduling, logistics tracking, manufacturing status updates, and treatment center coordination into unified systems. Real-time visibility enables proactive exception management rather than reactive firefighting. Chain of identity systems using biometric verification, RFID tags, and barcode scanning ensure product traceability from apheresis through infusion. Temperature monitoring, route optimization, and automated documentation reduce errors while accelerating vein-to-vein timelines by 15-30%.
- What role does real-world data play in cell and gene therapy development?
Real-world data from electronic health records, claims databases, and patient registries provides insights into long-term safety, effectiveness in diverse populations, and comparative treatment outcomes beyond controlled trial settings. The FDA is developing frameworks to incorporate real-world evidence into regulatory decisions, particularly for post-approval safety monitoring, label expansions, and rare disease indications where traditional trials face enrollment challenges. Standardized data formats like HL7 FHIR enable efficient real-world data integration into development programs.
Conclusion
Digital transformation represents more than technology adoption—it’s fundamental reimagining of how cell and gene therapies are developed, manufactured, and delivered to patients.
From AI-powered treatment prediction to automated manufacturing platforms, digital tools address critical challenges limiting CGT scalability and accessibility. Organizations implementing these technologies report faster development timelines, improved product quality, reduced costs, and better patient outcomes.
Yet technology alone doesn’t guarantee success. Effective transformation requires strategic vision, cross-functional collaboration, regulatory alignment, change management, and continuous optimization. Organizations must balance innovation with compliance, speed with quality, and automation with human oversight.
The FDA’s support for advanced manufacturing technologies through designation programs and enhanced engagement creates favorable regulatory environments for innovation. As digital capabilities mature and adoption expands, cell and gene therapy will increasingly deliver on its promise of curative treatments for previously untreatable conditions.
Now is the time for CGT organizations to assess digital maturity, identify high-value opportunities, and begin transformation journeys. The competitive advantages—and patient benefits—make digital transformation not just beneficial but essential.
Ready to advance your organization’s digital transformation? Start by evaluating current capabilities against industry benchmarks, identifying pain points with measurable business impact, and prioritizing initiatives delivering quick wins while building toward comprehensive change.


