Quick Summary: Digital transformation for bioprocessing combines AI, digital twins, real-time data analytics, and hybrid modeling to revolutionize biomanufacturing. According to market research (e.g., Fortune Business Insights), the global artificial intelligence market size is projected to grow from $294.16 billion in 2025 to $1771.62 billion by 2032, exhibiting a CAGR of 29.2%. These technologies enable manufacturers to optimize cell culture processes, accelerate batch release, reduce development costs, and maintain regulatory compliance in an increasingly complex production environment.
The biopharmaceutical industry faces a critical crossroads. With drug candidate attrition rates at 96% and average development costs of over $3 billion, manufacturers can’t afford to rely on traditional approaches. Digital transformation isn’t just another buzzword—it’s becoming the fundamental operating system for modern bioprocessing.
Here’s the thing though: implementing digital solutions in bioprocessing isn’t as straightforward as plugging in new software. Manufacturing environments generate massive amounts of data, but most organizations struggle to turn that information into actionable insights.
This guide breaks down exactly how digital technologies are reshaping bioprocessing, which tools actually deliver results, and what manufacturers need to know to stay competitive.
Why Digital Transformation Matters Now
The bioprocessing landscape has changed dramatically. Generative AI adoption in biopharma has reached 54% uptake by 2025, according to life sciences industry trends. But adoption alone doesn’t guarantee success.
Traditional manufacturing relied on manual data collection, periodic sampling, and retrospective batch analysis. That approach creates several problems:
- Batch deviations go undetected until it’s too late to correct
- Process optimization happens slowly through trial and error
- Scale-up failures waste time and resources
- Regulatory documentation becomes a bottleneck
Real talk: these limitations directly impact the bottom line. Monoclonal antibody purification processes typically achieve 70% product recovery with purity exceeding 95%, according to research published in Biotechnology and Bioengineering. Yet many manufacturers leave significant yield on the table because they can’t identify optimization opportunities in real time.
Core Technologies Driving Transformation
Several digital technologies are proving their value in bioprocessing environments. Each addresses specific challenges in the manufacturing workflow.
Digital Twins and Virtual Modeling
Digital twins create virtual representations of physical bioprocessing systems. These models simulate how changes in process parameters affect outcomes before implementing them in production.
Research published in the International Journal of Pharmaceutics highlights how digital twins reduce risk from drug discovery through continuous manufacturing. The technology allows manufacturers to test scenarios virtually, identifying potential issues before they impact actual production batches.
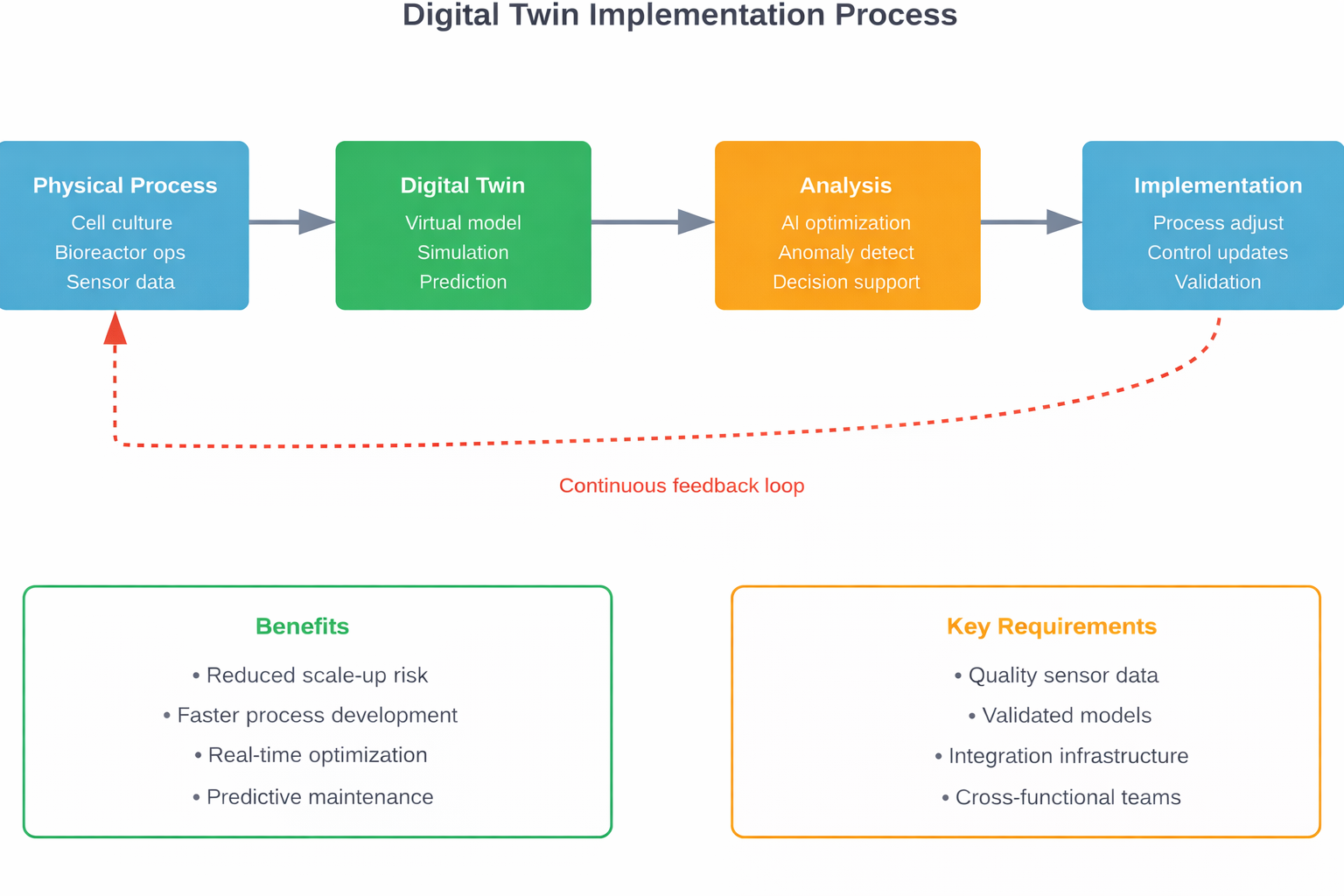
The most advanced CHO cell models now include 3,597 genes, 11,004 reactions, and 7,377 metabolites, according to research in Computational and Structural Biotechnology Journal. This level of detail enables precise metabolic predictions that weren’t possible with simpler models.
Real-Time Data Analytics and PAT
Process Analytical Technology allows continuous monitoring throughout manufacturing. Instead of waiting for offline lab results, PAT systems provide immediate feedback on critical quality attributes.
Data-defined bioprocesses take this further by creating seamless data flow across systems. This enables AI to continuously optimize operations while making analytical decisions automatically.
One global vaccine manufacturer applied these principles to improve yield based on approximately 10 years of manufacturing history covering thousands of parameters. The system automatically generates real-time reports, speeding up batch release by enabling review by exception rather than comprehensive manual checks.
Hybrid Modeling Approaches
Hybrid models combine mechanistic understanding with machine learning. The mechanistic component captures known biological and chemical principles. Machine learning fills gaps where fundamental understanding remains incomplete.
This approach proves particularly valuable for complex bioprocesses where pure mechanistic models become unwieldy and pure ML models lack interpretability. Hybrid models balance both needs effectively.
Implementing Digital Solutions
Technology selection matters less than implementation strategy. Many digital transformation initiatives fail not because of poor tools, but because of inadequate planning and change management.
Start With Quality by Design Principles
Quality by Design establishes the foundation for digital bioprocessing. QbD identifies critical process parameters and quality attributes before selecting digital tools to monitor and control them.
The FDA’s Current Good Manufacturing Practice regulations emphasize process understanding and control. Digital technologies support compliance by providing continuous documentation and real-time process monitoring.
| QbD Element | Digital Technology Support | Primary Benefit |
|---|---|---|
| Design space definition | Digital twins, DoE software | Faster optimization |
| Critical parameter monitoring | PAT sensors, real-time analytics | Immediate deviation detection |
| Process understanding | Hybrid models, AI analysis | Deeper mechanistic insights |
| Control strategy | Automated control systems | Consistent quality |
| Continuous improvement | Data lakes, ML algorithms | Ongoing optimization |
Build Data Infrastructure First
Sophisticated analytics require quality data. But wait—that means infrastructure investments come before algorithm development.
Key infrastructure components include:
- Standardized data formats across instruments and systems
- Secure data storage with appropriate retention policies
- Integration platforms connecting disparate manufacturing systems
- Version control for process parameters and models
Research in MAbs journal emphasizes unified digital platforms for data analysis and workflow management. Fragmented systems create data silos that undermine advanced analytics.
Address Regulatory Considerations Proactively
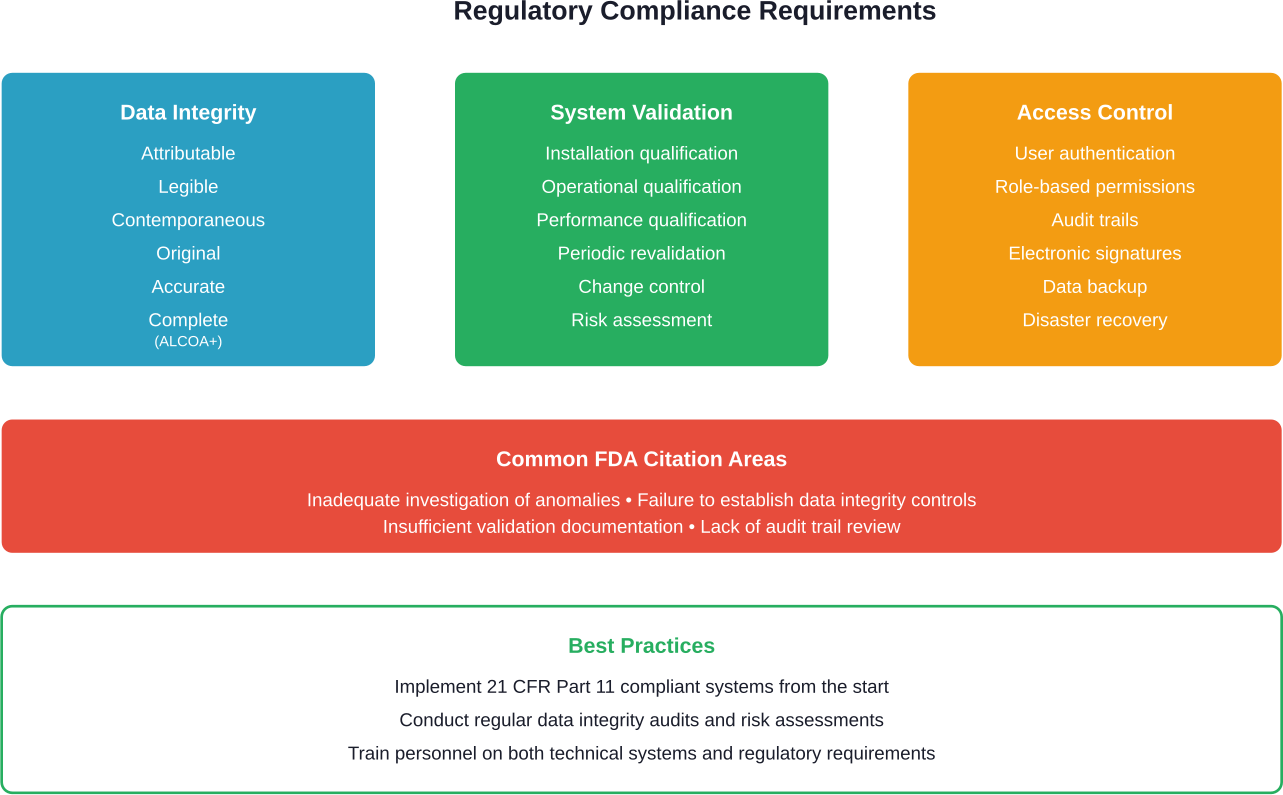
Digital systems must meet regulatory requirements for pharmaceutical manufacturing. This includes data integrity principles known as ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, plus complete, consistent, enduring, and available).
FDA warning letters frequently cite CGMP violations related to data integrity. Digital systems must be validated, with appropriate access controls, audit trails, and change management procedures.
Modernize Bioprocessing Infrastructure With the Right Support
Bioprocessing companies often deal with disconnected systems, legacy software, and complex data environments that slow down production and analysis. Digital transformation focuses on upgrading core platforms, connecting lab and manufacturing systems, and improving how operational data flows across teams.
A-listware supports organizations that need to modernize their technology stack. Their engineers help review existing infrastructure, upgrade legacy systems, and implement scalable software or cloud environments that better support production and research workflows.
If your bioprocessing systems need a stable digital foundation, bring in A-listware to help plan and implement the transition.
Continuous Manufacturing and Process Intensification
Continuous manufacturing represents a fundamental shift from batch production. This approach reduces facility footprint, improves consistency, and enables real-time quality assurance.
But here’s the catch: continuous processes generate exponentially more data than batch operations. Without digital systems to manage that complexity, the operational burden becomes overwhelming.
Process Analytical Technology becomes essential rather than optional in continuous manufacturing. Real-time monitoring and control keep processes within specifications without manual intervention.
Research in Biotechnology and Bioengineering notes that monoclonal antibody purification typically targets less than 100 ppm host cell protein, less than 10 ng per dose host cell DNA, and product purity exceeding 95%. Continuous processes with integrated PAT maintain these specifications more consistently than batch operations.
AI and Machine Learning Applications
Artificial intelligence adds predictive and optimization capabilities to bioprocessing. The technology has moved beyond pilot projects into production environments at leading manufacturers.
Predictive Analytics for Process Optimization
Machine learning algorithms identify patterns in historical manufacturing data that humans miss. These patterns reveal relationships between process parameters and product quality attributes.
Predictive models forecast batch outcomes based on early process indicators. This enables corrective action before quality issues develop, reducing batch failures and improving yield.
Anomaly Detection and Real-Time Alerts
AI systems continuously monitor process parameters, flagging deviations from normal operating ranges. Unlike simple threshold alerts, ML-based anomaly detection accounts for complex parameter interactions and subtle drift.
This proves particularly valuable for identifying equipment issues before they impact product quality. Predictive maintenance reduces unplanned downtime and extends equipment life.
| AI Application | Implementation Complexity | Typical ROI Timeline |
|---|---|---|
| Predictive batch outcomes | Medium | 6-12 months |
| Real-time anomaly detection | Medium-High | 3-9 months |
| Process optimization | High | 12-24 months |
| Automated batch release | High | 18-36 months |
| Predictive maintenance | Medium | 6-18 months |
Overcoming Implementation Challenges
Digital transformation faces predictable obstacles. Addressing these proactively increases success probability.
Data Quality and Availability
Many organizations discover their historical data isn’t suitable for advanced analytics. Inconsistent formats, missing metadata, and data gaps limit model training.
Starting with prospective data collection—even before implementing advanced analytics—builds the foundation for future initiatives. Clean, well-organized data becomes an asset that appreciates over time.
Skills and Organizational Change
Digital bioprocessing requires cross-functional collaboration between process engineers, data scientists, quality professionals, and IT specialists. These groups often speak different languages and have different priorities.
Successful organizations create integrated teams with shared objectives. Training programs help traditional manufacturing personnel develop data literacy while teaching data scientists about bioprocessing fundamentals.
Integration With Legacy Systems
Most facilities operate a mix of modern and legacy equipment. Legacy systems may lack digital connectivity or use proprietary data formats.
Middleware platforms bridge these gaps, extracting data from legacy systems and converting it to standardized formats. While not ideal, this approach enables digital transformation without replacing functional equipment prematurely.
Measuring Success and ROI
Digital initiatives require clear success metrics. Financial justification remains important, but leading organizations also track operational and quality improvements.
Key performance indicators include:
- Batch yield improvement and reduction in process variability
- Faster development timelines from concept to commercial production
- Reduced batch failures and investigation cycles
- Improved equipment utilization and reduced downtime
- Faster batch release through automated data review
The estimated average cost to develop a new drug was approximately $2.6 billion (in 2013 dollars), but when adjusted for inflation by 2026, this figure exceeds $3 billion.
Future Directions
Digital bioprocessing continues evolving rapidly. Several emerging trends deserve attention.
Multimodal AI systems integrate diverse data types—genomic sequences, protein structures, process parameters, and product quality data. This holistic approach reveals relationships invisible when analyzing data types in isolation.
Edge computing brings advanced analytics closer to manufacturing equipment. This reduces latency for real-time control while addressing data security concerns about cloud connectivity.
Personalized medicine creates unique manufacturing challenges. Digital tools enable flexible production systems that can efficiently manufacture small batches of patient-specific therapies.
Frequently Asked Questions
- What is digital transformation in bioprocessing?
Digital transformation in bioprocessing refers to integrating advanced technologies like AI, digital twins, real-time analytics, and automated control systems into biomanufacturing operations. This enables data-driven decision making, process optimization, and continuous improvement rather than relying solely on traditional manual approaches and batch-based quality control.
- How do digital twins improve bioprocess development?
Digital twins create virtual models of bioprocessing systems that simulate how parameter changes affect outcomes before implementation. This reduces scale-up risk, accelerates process development, and enables optimization through virtual experimentation. Research shows digital twins can include thousands of metabolic reactions and genetic elements, providing detailed predictions of cell culture behavior.
- What are data-defined bioprocesses?
Data-defined bioprocesses use real-time data flow integrated across systems with AI continuously optimizing operations and making analytical decisions. Instead of periodic manual sampling and offline analysis, these systems provide immediate feedback on process performance, enabling faster corrective action and automated batch release through exception-based review.
- How does PAT support digital bioprocessing?
Process Analytical Technology provides continuous monitoring of critical process parameters and quality attributes throughout manufacturing. PAT generates real-time data that feeds digital twins, AI optimization algorithms, and automated control systems. This enables immediate deviation detection and response rather than discovering issues only during end-of-batch testing.
- What regulatory considerations apply to digital bioprocessing systems?
Digital systems must comply with FDA Current Good Manufacturing Practice regulations including data integrity requirements. Systems need validation documentation, audit trails, access controls, and electronic signature capabilities. The FDA emphasizes that digital tools should enhance process understanding and control while maintaining data that is attributable, legible, contemporaneous, original, and accurate.
- What skills are needed for digital bioprocessing implementation?
Successful implementation requires cross-functional teams combining bioprocess engineering knowledge, data science expertise, quality system understanding, and IT infrastructure capabilities. Organizations often need training programs to develop data literacy among traditional manufacturing personnel while teaching data scientists about bioprocessing fundamentals and regulatory requirements.
- What ROI can organizations expect from digital bioprocessing initiatives?
Return on investment varies by application and implementation quality. Predictive analytics for batch outcomes typically show ROI within 6-12 months through reduced batch failures and improved yield. Process optimization initiatives may require 12-24 months but generate ongoing value. Financial benefits come from improved yield, faster development, reduced downtime, and accelerated batch release.
Conclusion
Digital transformation fundamentally changes how bioprocessing works. The technologies aren’t speculative anymore—AI, digital twins, and real-time analytics are delivering measurable results at leading manufacturers.
But success requires more than technology adoption. Organizations need data infrastructure, cross-functional collaboration, regulatory compliance frameworks, and clear implementation strategies. Starting with focused pilot projects in high-value areas builds capability while demonstrating ROI.
The competitive landscape demands continuous improvement. Manufacturers that effectively leverage digital tools gain advantages in speed, efficiency, and quality that become difficult for competitors to match.
Ready to transform your bioprocessing operations? Start by assessing your current data infrastructure and identifying high-impact use cases where digital solutions can deliver quick wins. Build from there with a clear roadmap that balances ambition with practical implementation considerations.


